Skin, like all body tissue, is made up of cells. The two main cell types in the outermost layer of skin (the epidermis) are called keratinocytes and melanocytes. There are several layers of epidermal keratinocytes with the lowest level being known as the basal layer, and the main viable (live) layer composed of squamous cells. Melanocytes produce the skin pigment melanin. Different skin cancers are named after the skin cell that the cancer resembles or develops from - basal cell carcinoma, squamous cell carcinoma, and melanoma.
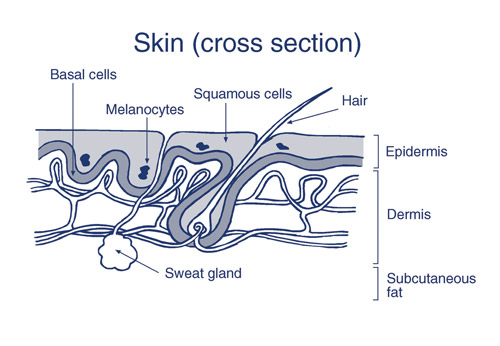
Figure 1: Skin cross-section showing basal cells, squamous cells and melanocytes
Melanoma
Aetiology
Melanoma is the least common skin cancer but it is also the most dangerous.[1] Melanoma is the result of unrepaired DNA damage to melanocytes. This triggers abnormal cell growth[2][3][4][5] attributable to a combination of genetic risk factors and overexposure to ultraviolet (UV) radiation.[6]
Types of melanoma
Cutaneous (malignant) melanomas are classified into four types by histological appearance and biological behaviour (the way they look under the microscope, how thick they are, how far and fast they’ve spread). However, there are melanomas that are of unclassifiable histogenetic type.[7][8] It has been argued that not only histological appearance but the particular "biochemical and genetic aberrations (abnormalities) correlating with anatomic (body) site and amount of cutaneous (skin) exposure to sunlight" should also be taken into account when classifying tumours.[8][9]
Melanoma may arise from a pre-existent melanocytic naevus (mole), but can also grow de novo on the skin, without a pre-existing pigmented lesion. A carcinoma in situ is an early form of cancer that is not yet invasive. In the case of melanoma, in situ means that abnormal skin cells are contained within the epidermis and have not penetrated deeper into the dermis.
- Superficial spreading melanoma (SSM): SSM typically forms in areas intermittently exposed to the sun, and remains in the upper layers of the skin (radial growth phase) for a long time before invading more deeply (vertical growth phase). It appears as an irregular spreading growth of discoloured skin that is variegated in colour[7] usually dominated by brown or black.[10] SSM is most common among younger individuals.[7] SSM is the most common subtype of melanoma (except in patients with extensive sun exposure[11]) and accounts for approximately 55-60% melanoma in Australia,[12] but only 30% of deaths.[13]
- Lentigo maligna melanoma (LMM): Lentigo maligna (LM) arises on chronically sun-exposed/sun-damaged skin, on the head or neck of patients – usually among those who are over 40 years of age.[14] Lesions are typically asymmetrical, with an uneven colour and border, enlarging and changing in appearance over time. There are also rare morphologically divergent clinical variants lacking these classical features of LM.[9] LM may evolve into an invasive form (lentigo maligna melanoma—LMM) over the course of many years, or not at all.[9] LMM accounts for approximately 10-15% of cases in Australia.[12]
- Acral lentiginous melanoma (ALM): This subtype occurs almost exclusively on hands and feet. ALM spreads superficially for months to years before becoming invasive. However ALM can advance more rapidly than SSM or LMM. It is most common among those of African descent and Asians, least common among Caucasians.[7][15] This subtype makes up 1-2% of melanomas in Australia.[12]
- Nodular melanoma (NM): Compared with other melanomas with radial growth, NM are relatively small in diameter, symmetrical in shape, uniform in colour, and frequently amelanotic (i.e. lacking pigment).[16] Nodular melanoma is usually invasive at the time of diagnosis and constitutes nearly half of lesions greater than 2mm in thickness. Nodular melanomas comprise 10-15% of melanomas diagnosed in Australia.[12] Nodular melanoma contributes highly to melanoma mortality[13] due to its association with increased growth rate and thickness at diagnosis.[13][16]
- Desmoplastic melanoma: This subtype accounts for 1-2% of cases in Australia and are characterized by malignant spindled melanocytes with surrounding fibrous stroma. They can be difficult to diagnose both clinically and on histopathology.[12]
Other subtypes include malignant melanoma NOS (Not Otherwise Specified), balloon cell melanoma, malignant melanoma regressing, amelanotic melanoma and malignant melanoma in junctional naevus, among others.[17]
Current melanoma subtype classifications are less useful for classifying paediatric melanoma; a retrospective study of melanomas diagnosed before 20 years of age found that 44% were ‘histopathologically unclassifiable using current melanoma subtypes’. The authors suggest an adapted, paediatric-specific 'ABCDE' criteria be used in conjunction with the standard in order to identify lesions as cancerous.[18]
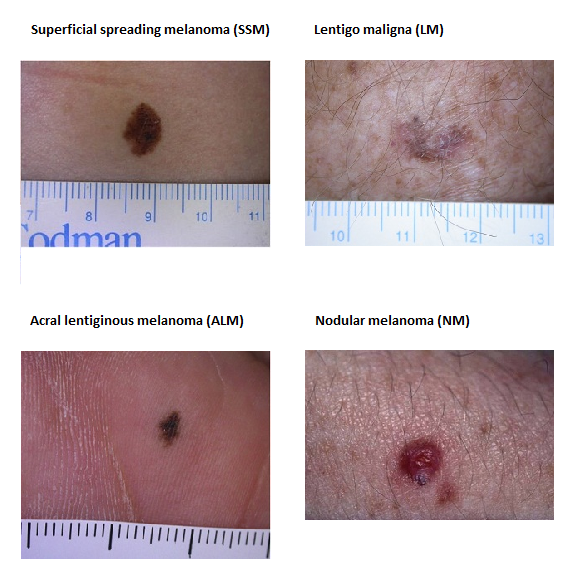
Figure 2: Typical presentation of the four main types of cutaneous melanoma
SMM, LM, ALM images generously provided by Dr Alvin Chong, Skin & Cancer Foundation Victoria; NM image generously provided by Associate Professor John Kelly, Victorian Melanoma Service, Alfred Health
Clinical stages
Malignant melanoma is a spectrum with highly curable, thin primary cutaneous melanoma on one end and less treatable, often deadly metastatic melanoma on the other.[3]
According to the current melanoma staging system, as recommended by the American Joint Committee on Cancer 2018,[19] tumours are classified based on prognostic factors. Factors include tumour Breslow thickness, the appearance of ulceration, and mitotic rate (speed of cancerous cell-division). Clarke’s level of invasion is considered when the mitotic rate cannot be determined. Of these factors, Breslow’s thickness is the most important in determining prognosis for invasive melanomas, and is used to determine tumour severity for the early (prior to metastasis) stages 0-II.
Clinical stages are classified as 0-IV; from non-invasive to invasive.
Stage 0 (melanoma in situ) is when the tumour is confined to the epidermis. Stage I-II tumours are >4mm thick with or without ulceration. Stage III tumours have developed in-transit metastasis, spreading more than 2cm from the primary tumour site through a lymph vessel, or have metastasised to a local lymph node. Stage IV means the tumour has metastasised to distant skin sites, lymph nodes or internal organs such as the lungs, liver, and brain.[19]
Breslow’s thickness (depth)
Breslow’s thickness (or depth) classifies tumours as thin melanomas (≤1mm), intermediate (>1-4mm), and thick (>4mm) according to the distance between the “upper (granular) layer of epidermis and deepest point of tumour penetration”.[20] Tumour thickness is more predictive of prognosis than the level of tumour invasion.[20] Thin lesions ≤1mm in thickness are treated through surgery and have a high, almost 100% (depending on presence of ulceration), five-year survival in Australia.[21] In contrast, survival for lesions >4mm thick is only 54%.[21]
Nodular melanoma and increasing age are significantly associated with melanoma thickness.[16][22] The median thickness for nodular melanoma was 2.6mm at diagnosis compared with 0.6mm for superficial spreading melanoma.[13] Due to its association with tumour thickness and hence mortality, nodular melanoma is the largest contributor to melanoma mortality within Victoria.[13]
Incidence of thin melanomas (≤1mm) in Australia increased between 1990 to 1996, and stabilised between 1996 and 2006. There was a promising decrease in the incidence of thin melanomas among younger people - likely the result of skin cancer awareness campaigns. Frequency of thin melanomas within this 17 year period was 65.4% for females and 60.5% for males (62.7% total) of all invasive cutaneous melanomas diagnosed within this period.[23]
However, invasive thick melanoma (>4mm) incidence increased up to 2006, peaking at a total of 5.9% of invasive cutaneous melanomas diagnosed in 2004 and 2006. Between 1990 and 2006, invasive thick melanoma accounted for 4.3% of invasive melanoma diagnosed in females and 5.7% of invasive melanoma in males. Within the period 1996 to 2006 the increase in thick melanomas was evident in most male age groups, but was only evident among females aged more than 75 years. Incidence of thick melanomas was highest in northern latitudes; however the magnitude of increase was greatest in southern states/territories. The increase in thick melanomas does not appear to have adversely affected mortality rates, Baade and colleagues speculate that this may be, in part, due to an increase in detecting lesions before they metastasise and is therefore consistent with the observed decrease in retroactively classifying metastasised melanomas as being of ‘unknown thickness’ at diagnosis.[23]
Non-melanocytic skin cancer
Non-melanocytic skin cancers (NMSC), also known as keratinocyte carcinomas, are cancers of the epithelial layer (epidermis). They are more common than melanoma, with incidence of NMSC in Australia more than five times the incidence of all other cancers combined in 2002, the highest in the world.[24][25] According to Medicare, there were 1,107,837 NMSC treatments in Australia in 2022.[26] It is estimated that 69% of Australians (73% of men, 65% of women) will have at least one excision for histologically confirmed keratinocyte cancer in their lifetime (60% to age 79 years).[27] Basal cell carcinoma (BCC) is the most common NMSC.[25]
Aetiology
As in the case of melanoma, UV exposure (chronic exposure for SCC and BCC, intermittent exposure for BCC) is the main environmental risk factor for NMSC.[28][29][30][31][32] However, the exact role of UV exposure (amount, timing, and pattern of exposure) is yet to be determined,[33] particularly for BCC.[34] As with melanoma, there are also host risk factors that increase risk of developing NMSC.[35][36]
Basal cell carcinoma
Basal cell carcinoma (BCC) is the most common malignancy in humans and makes up approximately 70% of non-melanocytic skin cancers within Australia.[24] BCCs are common on areas which typically receive high or intermittent sun exposure.[37] Within Australia, BCC is more common in men than women, with the majority of BCCs appearing on the head and neck, followed by the trunk.[24]
BCC appears as malignant neoplasms (abnormal tissue growth) of basal appearing cells, which may develop as a pearly pink or flesh-coloured papule (small lump) or a scaly/dry area that is pale pink to red in colour, giving the appearance of an unhealed sore. BCCs may bleed and become inflamed, and dead tissue may slough off (ulcerate),[38] though they are usually asymptomatic.[36]
BCC rarely metastasises to become life-threatening,[39] unless it is left untreated and allowed to develop into a high-risk tumour larger than 2cm in diameter or 4mm in thickness.[36][40] Other consequences include the damage/destruction of surrounding tissue, and increasing difficulty of complete surgical excision, thereby increasing the chance of recurrence.[38] If treated with the ‘gold standard’ treatment—Mohs micrographic surgery (MMS)— the 5-year cure rate in Australia is high at 98.6% for excision of a primary BCC.[41]
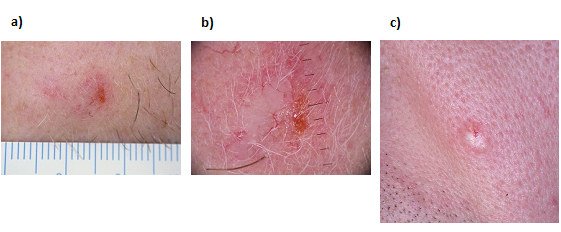
Figure 3: Typical presentation of BCC a) macro view of ulcerated superficial BCC (sBCC) b) dermatoscopy view of ulcerated sBCC c) macro view of nodular ulcerated BCC
Images generously provided by Dr Alvin Chong, Skin & Cancer Foundation Victoria
Squamous cell carcinoma
Squamous cell carcinoma (SCC) makes up approximately 30% of non-melanocytic skin cancers.[24] SCC usually appears on parts of the body most often exposed to the sun such as the head and neck, arms and legs.[24][37] A SCC appears as a thickened, red, scaly spot and may be tender to the touch, though usually SCCs are asymptomatic. SCC may become crusted or ulcerated.[36] Unlike BCC which develops de novo (in the absence of a precursor lesion), SCCs typically arise from existent pre-malignant, precursor lesions (actinic or solar keratosis, and Bowen's disease).[34][40] However, de novo SCC can occur and is an aggressive, distinct clinical variant.[42] SCC may also arise from chronic ulcers or scars.[36]
SCC may grow more rapidly than BCC.[36] Like BCC, SCC does not typically metastasise but requires treatment to prevent tumour growth.[42] Metastasis, when it occurs, is usually to regional lymph nodes, and is more common for SCC arising on the lips or ears. If treated with MMS, the gold standard treatment, the five-year cure rate is 97.4%.[43]
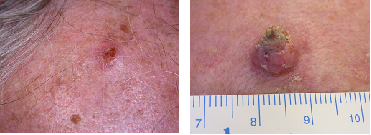
Figure 4: Two examples of SCC
Images generously provided by Dr Alvin Chong, Skin & Cancer Foundation Victoria
- Marks R. An overview of skin cancers. Incidence and causation. Cancer 1995 Jan 15;75(2 Suppl):607-12 Available from: http://www.ncbi.nlm.nih.gov/pubmed/7804986.
- Gilchrest BA, Eller MS. DNA photodamage stimulates melanogenesis and other photoprotective responses. J Investig Dermatol Symp Proc 1999 Sep;4(1):35-40 Available from: http://www.ncbi.nlm.nih.gov/pubmed/10537005.
- Markovic SN, Erickson LA, Rao RD, Weenig RH, Pockaj BA, Bardia A, et al. Malignant melanoma in the 21st century, part 1: epidemiology, risk factors, screening, prevention, and diagnosis. Mayo Clin Proc 2007 Mar;82(3):364-80 Available from: http://www.ncbi.nlm.nih.gov/pubmed/17352373.
- Leiter U, Garbe C. Epidemiology of melanoma and nonmelanoma skin cancer--the role of sunlight. Adv Exp Med Biol 2008;624:89-103 Available from: http://www.ncbi.nlm.nih.gov/pubmed/18348450.
- Rigel DS. Epidemiology of melanoma. Semin Cutan Med Surg 2010 Dec;29(4):204-9 Available from: http://www.ncbi.nlm.nih.gov/pubmed/21277533.
- Jhappan C, Noonan FP, Merlino G. Ultraviolet radiation and cutaneous malignant melanoma. Oncogene 2003 May 19;22(20):3099-112 Available from: http://www.ncbi.nlm.nih.gov/pubmed/12789287.
- Porras BH, Cockerell CJ. Cutaneous malignant melanoma: classification and clinical diagnosis. Semin Cutan Med Surg 1997 Jun;16(2):88-96 Available from: http://www.ncbi.nlm.nih.gov/pubmed/9220547.
- Duncan LM. The classification of cutaneous melanoma. Hematol Oncol Clin North Am 2009 Jun;23(3):501-13, ix Available from: http://www.ncbi.nlm.nih.gov/pubmed/19464599.
- Reed JA, Shea CR. Lentigo maligna: melanoma in situ on chronically sun-damaged skin. Arch Pathol Lab Med 2011 Jul;135(7):838-41 Available from: http://www.ncbi.nlm.nih.gov/pubmed/21732771.
- Warycha MA, Christos PJ, Mazumdar M, Darvishian F, Shapiro RL, Berman RS, et al. Changes in the presentation of nodular and superficial spreading melanomas over 35 years. Cancer 2008 Dec 15;113(12):3341-8 Available from: http://www.ncbi.nlm.nih.gov/pubmed/18988292.
- Forman SB, Ferringer TC, Peckham SJ, Dalton SR, Sasaki GT, Libow LF, et al. Is superficial spreading melanoma still the most common form of malignant melanoma? J Am Acad Dermatol 2008 Jun;58(6):1013-20 Available from: http://www.ncbi.nlm.nih.gov/pubmed/18485983.
- Mar V, Teddy L, Kelly, J, Chamberlain A, Murray B, Cancer Council Australia Melanoma Guidelines Working Party. Clinical question: What are the clinical features of an atypical melanoma? In: Clinical practice guidelines for the diagnosis and management of melanoma. [homepage on the internet] Sydney, Australia: Melanoma Institute Australia.; [cited 2022 Aug 5; updated 2018 May 17]. Available from: https://www.cancer.org.au/clinical-guidelines/skin-cancer/melanoma.
- Mar V, Roberts H, Wolfe R, English DR, Kelly JW. Nodular melanoma: a distinct clinical entity and the largest contributor to melanoma deaths in Victoria, Australia. J Am Acad Dermatol 2013 Apr;68(4):568-75 Available from: http://www.ncbi.nlm.nih.gov/pubmed/23182058.
- Smalberger GJ, Siegel DM, Khachemoune A. Lentigo maligna. Dermatol Ther 2008 Nov;21(6):439-46 Available from: http://www.ncbi.nlm.nih.gov/pubmed/19076621.
- Phan A, Touzet S, Dalle S, Ronger-Savlé S, Balme B, Thomas L. Acral lentiginous melanoma: a clinicoprognostic study of 126 cases. Br J Dermatol 2006 Sep;155(3):561-9 Available from: http://www.ncbi.nlm.nih.gov/pubmed/16911282.
- Chamberlain AJ, Fritschi L, Giles GG, Dowling JP, Kelly JW. Nodular type and older age as the most significant associations of thick melanoma in Victoria, Australia. Arch Dermatol 2002 May;138(5):609-14 Available from: http://www.ncbi.nlm.nih.gov/pubmed/12020221.
- Australian Institute of Health and Welfare. Melanoma subtypes diagnosed in 2008. Canberra, Australia: Australian Institute of Health and Welfare; 2013.
- Cordoro KM, Gupta D, Frieden IJ, McCalmont T, Kashani-Sabet M. Pediatric melanoma: results of a large cohort study and proposal for modified ABCD detection criteria for children. J Am Acad Dermatol 2013 Jun;68(6):913-25 Available from: http://www.ncbi.nlm.nih.gov/pubmed/23395590.
- Melanoma of the Skin In: American Joint Committee on Cancer. AJCC Cancer Staging Manual. 8th ed. New York: Springer; 2017. p. 563-585.
- Breslow A. Tumor thickness, level of invasion and node dissection in stage I cutaneous melanoma. Ann Surg 1975 Nov;182(5):572-5 Available from: http://www.ncbi.nlm.nih.gov/pubmed/1190862.
- Australian Institute of Health and Welfare. Skin cancer in Australia. Cat. no. CAN 96. Canberra, Australia: AIHW; 2016 [cited 2016 Oct 5] Available from: http://www.aihw.gov.au/publication-detail/?id=60129555786.
- Curchin DJ, Harris VR, McCormack CJ, Smith SD. Changing trends in the incidence of invasive melanoma in Victoria, 1985-2015. Med J Aust 2018 Apr 2;208(6):265-269 Available from: http://www.ncbi.nlm.nih.gov/pubmed/29614940.
- Baade P, Meng X, Youlden D, Aitken J, Youl P. Time trends and latitudinal differences in melanoma thickness distribution in Australia, 1990-2006. Int J Cancer 2012 Jan 1;130(1):170-8 Available from: http://www.ncbi.nlm.nih.gov/pubmed/21344376.
- Staples MP, Elwood M, Burton RC, Williams JL, Marks R, Giles GG. Non-melanoma skin cancer in Australia: the 2002 national survey and trends since 1985. Med J Aust 2006 Jan 2;184(1):6-10 Available from: http://www.ncbi.nlm.nih.gov/pubmed/16398622.
- Lomas A, Leonardi-Bee J, Bath-Hextall F. A systematic review of worldwide incidence of nonmelanoma skin cancer. Br J Dermatol 2012 May;166(5):1069-80 Available from: http://www.ncbi.nlm.nih.gov/pubmed/22251204.
- Services Australia. Medicare Item Reports. [homepage on the internet] Canberra Australia; 2023 [cited 2023 Mar 30]. Available from: http://medicarestatistics.humanservices.gov.au/statistics/mbs_item.jsp.
- Olsen CM, Pandeya N, Green AC, Ragaini BS, Venn AJ, Whiteman DC. Keratinocyte cancer incidence in Australia: a review of population-based incidence trends and estimates of lifetime risk. Public Health Res Pract 2022 Mar 10;32(1) Available from: http://www.ncbi.nlm.nih.gov/pubmed/35290995.
- Bauer A, Diepgen TL, Schmitt J. Is occupational solar ultraviolet irradiation a relevant risk factor for basal cell carcinoma? A systematic review and meta-analysis of the epidemiological literature. Br J Dermatol 2011 Sep;165(3):612-25 Available from: http://www.ncbi.nlm.nih.gov/pubmed/21605109.
- Vishvakarman D, Wong JC. Description of the use of a risk estimation model to assess the increased risk of non-melanoma skin cancer among outdoor workers in Central Queensland, Australia. Photodermatol Photoimmunol Photomed 2003 Apr;19(2):81-8 Available from: http://www.ncbi.nlm.nih.gov/pubmed/12945807.
- Schmitt J, Seidler A, Diepgen TL, Bauer A. Occupational ultraviolet light exposure increases the risk for the development of cutaneous squamous cell carcinoma: a systematic review and meta-analysis. Br J Dermatol 2011 Feb;164(2):291-307 Available from: http://www.ncbi.nlm.nih.gov/pubmed/21054335.
- Armstrong BK, Kricker A. The epidemiology of UV induced skin cancer. J Photochem Photobiol B 2001 Oct;63(1-3):8-18 Available from: http://www.ncbi.nlm.nih.gov/pubmed/11684447.
- Kricker A, Armstrong BK, English DR, Heenan PJ. Does intermittent sun exposure cause basal cell carcinoma? a case-control study in Western Australia. Int J Cancer 1995 Feb 8;60(4):489-94 Available from: http://www.ncbi.nlm.nih.gov/pubmed/7829262.
- Wong CS, Strange RC, Lear JT. Basal cell carcinoma. BMJ 2003 Oct 4;327(7418):794-8 Available from: http://www.ncbi.nlm.nih.gov/pubmed/14525881.
- Madan V, Lear JT, Szeimies RM. Non-melanoma skin cancer. Lancet 2010 Feb 20;375(9715):673-85 Available from: http://www.ncbi.nlm.nih.gov/pubmed/20171403.
- Telfer NR, Colver GB, Morton CA, British Association of Dermatologists. Guidelines for the management of basal cell carcinoma. Br J Dermatol 2008 Jul;159(1):35-48 Available from: http://www.ncbi.nlm.nih.gov/pubmed/18593385.
- Albert MR, Weinstock MA. Keratinocyte carcinoma. CA Cancer J Clin 2003 Sep;53(5):292-302 Available from: http://www.ncbi.nlm.nih.gov/pubmed/14570228.
- Youl PH, Janda M, Aitken JF, Del Mar CB, Whiteman DC, Baade PD. Body-site distribution of skin cancer, pre-malignant and common benign pigmented lesions excised in general practice. Br J Dermatol 2011 Jul;165(1):35-43 Available from: http://www.ncbi.nlm.nih.gov/pubmed/21443534.
- Crowson AN. Basal cell carcinoma: biology, morphology and clinical implications. Mod Pathol 2006 Feb;19 Suppl 2:S127-47 Available from: http://www.ncbi.nlm.nih.gov/pubmed/16446711.
- Roewert-Huber J, Lange-Asschenfeldt B, Stockfleth E, Kerl H. Epidemiology and aetiology of basal cell carcinoma. Br J Dermatol 2007 Dec;157 Suppl 2:47-51 Available from: http://www.ncbi.nlm.nih.gov/pubmed/18067632.
- Samarasinghe V, Madan V. Nonmelanoma skin cancer. J Cutan Aesthet Surg 2012 Jan;5(1):3-10 Available from: http://www.ncbi.nlm.nih.gov/pubmed/22557848.
- Leibovitch I, Huilgol SC, Selva D, Richards S, Paver R. Basal cell carcinoma treated with Mohs surgery in Australia II. Outcome at 5-year follow-up. J Am Acad Dermatol 2005 Sep;53(3):452-7 Available from: http://www.ncbi.nlm.nih.gov/pubmed/16112352.
- Cassarino DS, Derienzo DP, Barr RJ. Cutaneous squamous cell carcinoma: a comprehensive clinicopathologic classification--part two. J Cutan Pathol 2006 Apr;33(4):261-79 Available from: http://www.ncbi.nlm.nih.gov/pubmed/16630176.
- Leibovitch I, Huilgol SC, Selva D, Hill D, Richards S, Paver R. Cutaneous squamous cell carcinoma treated with Mohs micrographic surgery in Australia II. Perineural invasion. J Am Acad Dermatol 2005 Aug;53(2):261-6 Available from: http://www.ncbi.nlm.nih.gov/pubmed/16021121.

